Experiment description
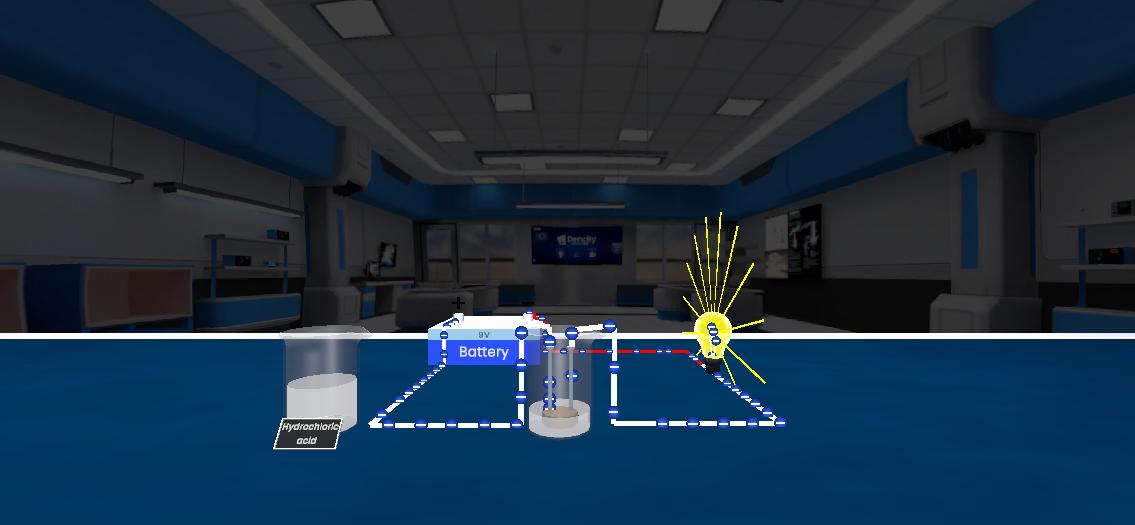
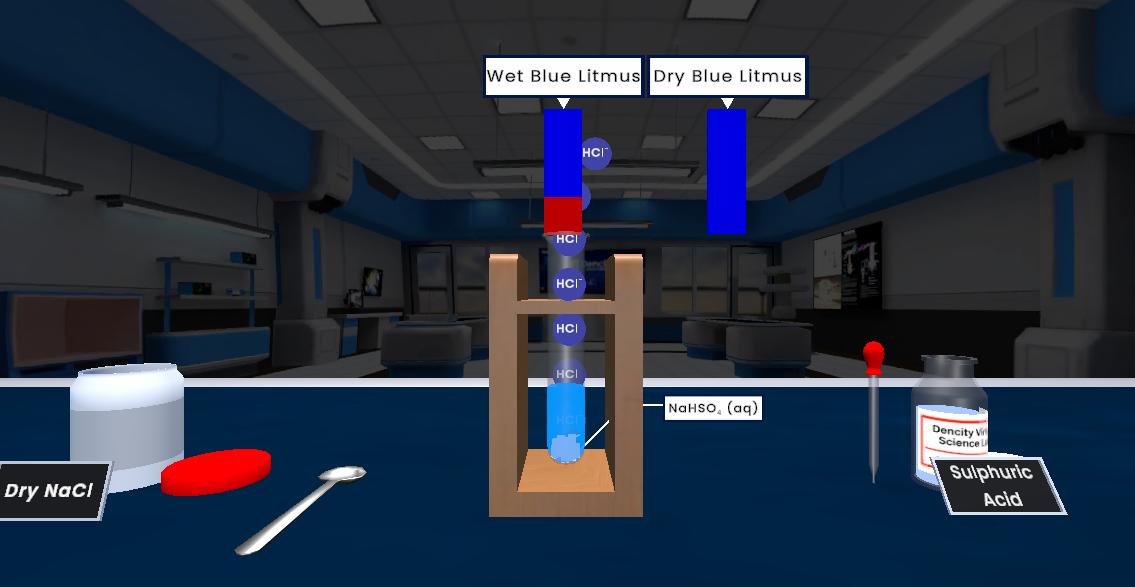
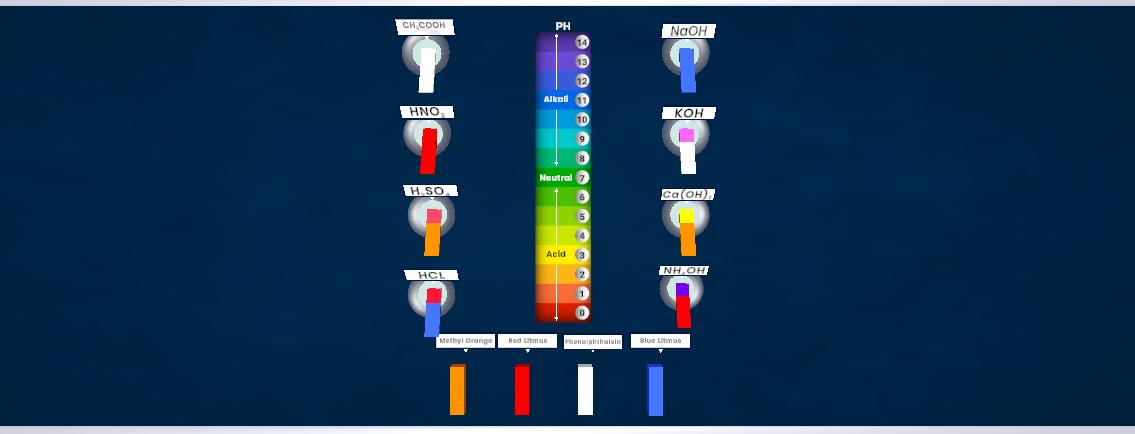
This experiment demonstrates electrical conductivity in acids, bases, and electrolytes. Different solutions are tested by dipping two electrodes connected to a bulb into the solution. If the solution contains free ions, it conducts electricity and the bulb glows, showing that the presence of charged particles allows electric current to pass.