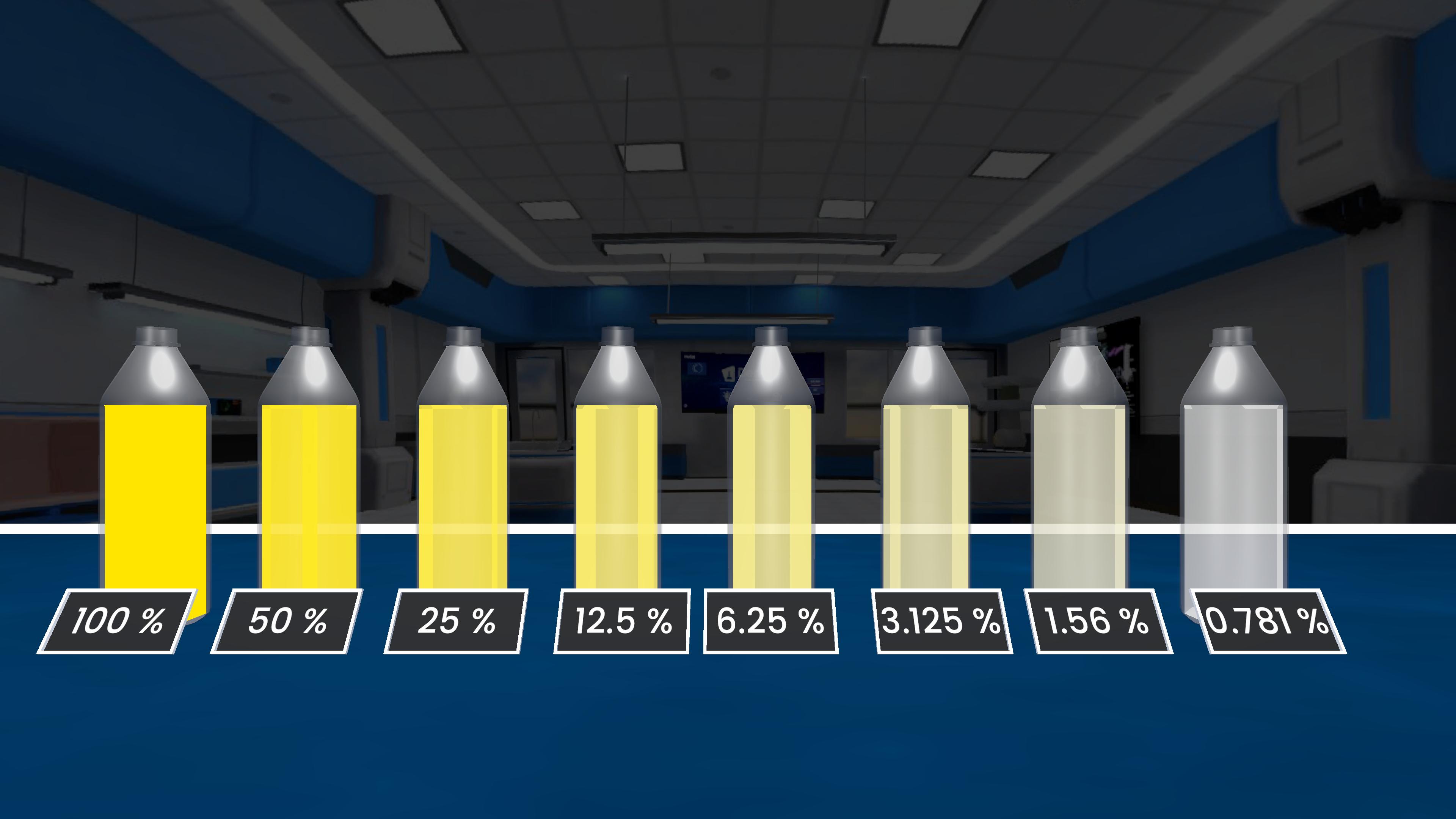
Experiment description
This experiment shows how concentration affects the rate of a chemical reaction. As the concentration of sodium dithionite increases, the reaction with hydrochloric acid happens faster. Beakers with higher concentration take less time to complete the reaction, proving that increasing reactant concentration increases the rate of reaction.