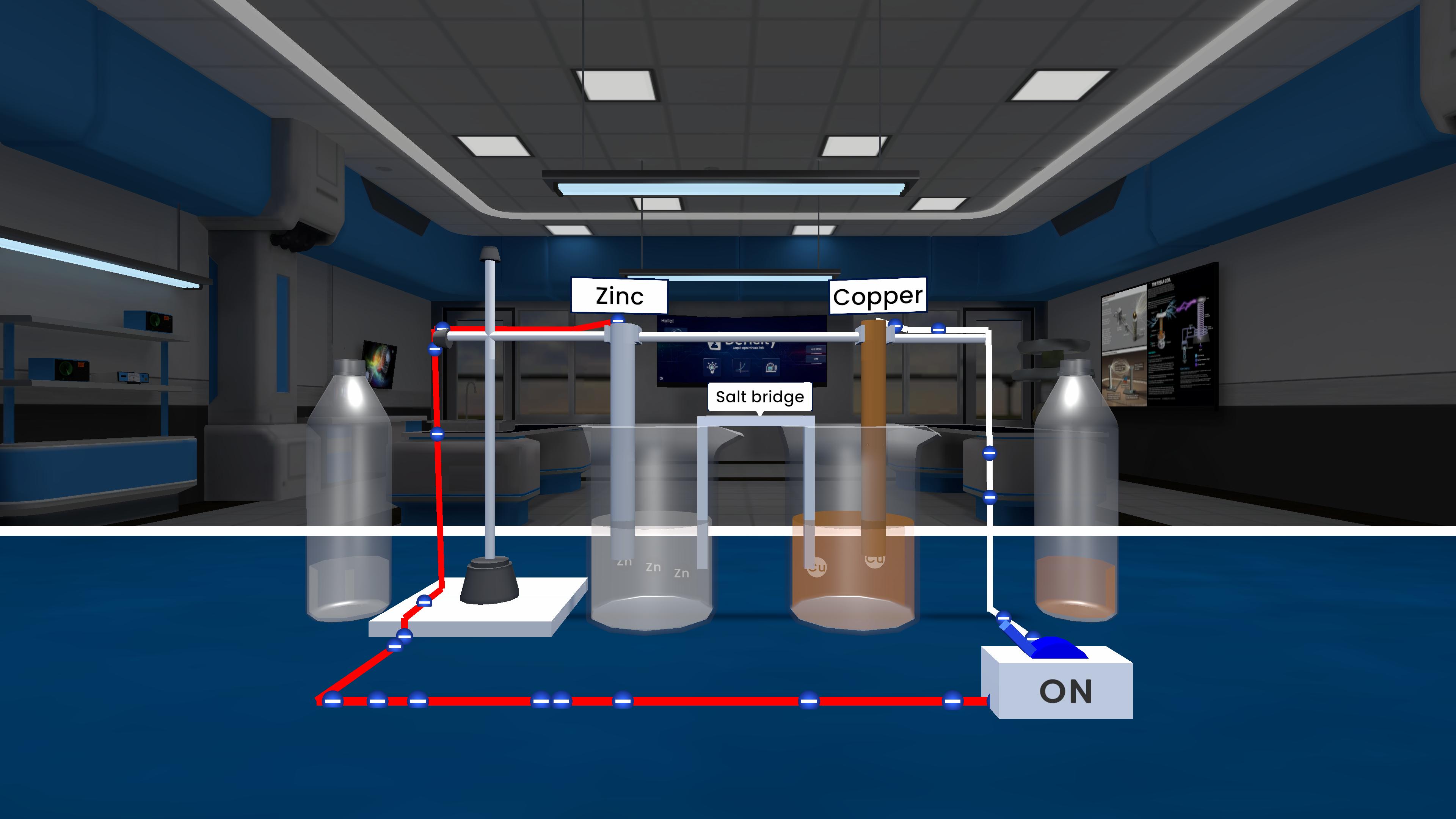
Experiment description
This experiment demonstrates how electrical energy is produced from a chemical reaction. Zinc acts as the anode and loses electrons, while copper acts as the cathode and gains electrons. A salt bridge allows ions to move between the solutions and keeps the circuit balanced, producing a steady flow of current.