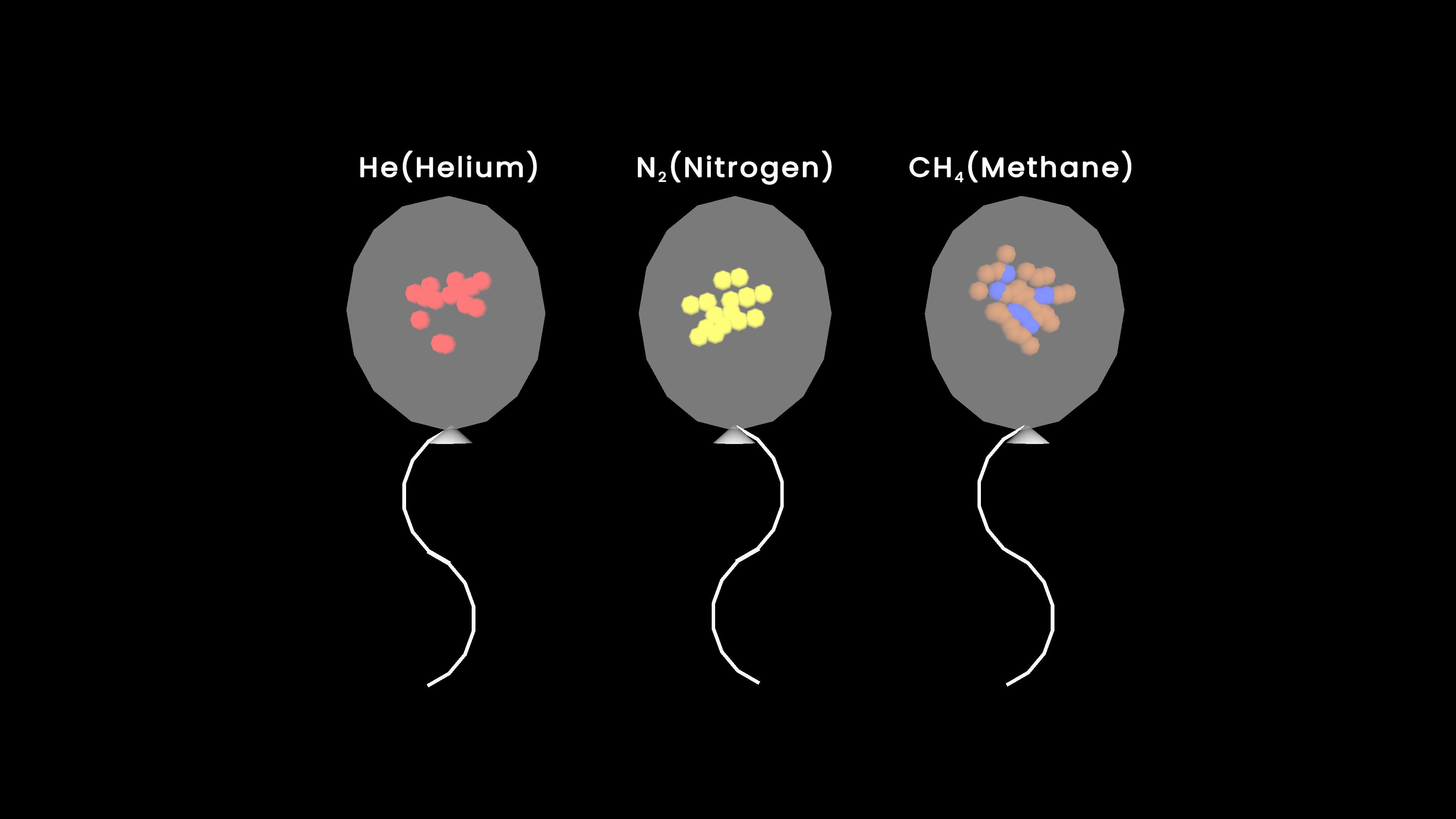
Experiment description
This experiment demonstrates Charles's Law, which states that the volume of a gas is directly proportional to its temperature at constant pressure. As the temperature changes, the volume of the gas changes accordingly, showing how gases expand when heated and contract when cooled.