Experiment description
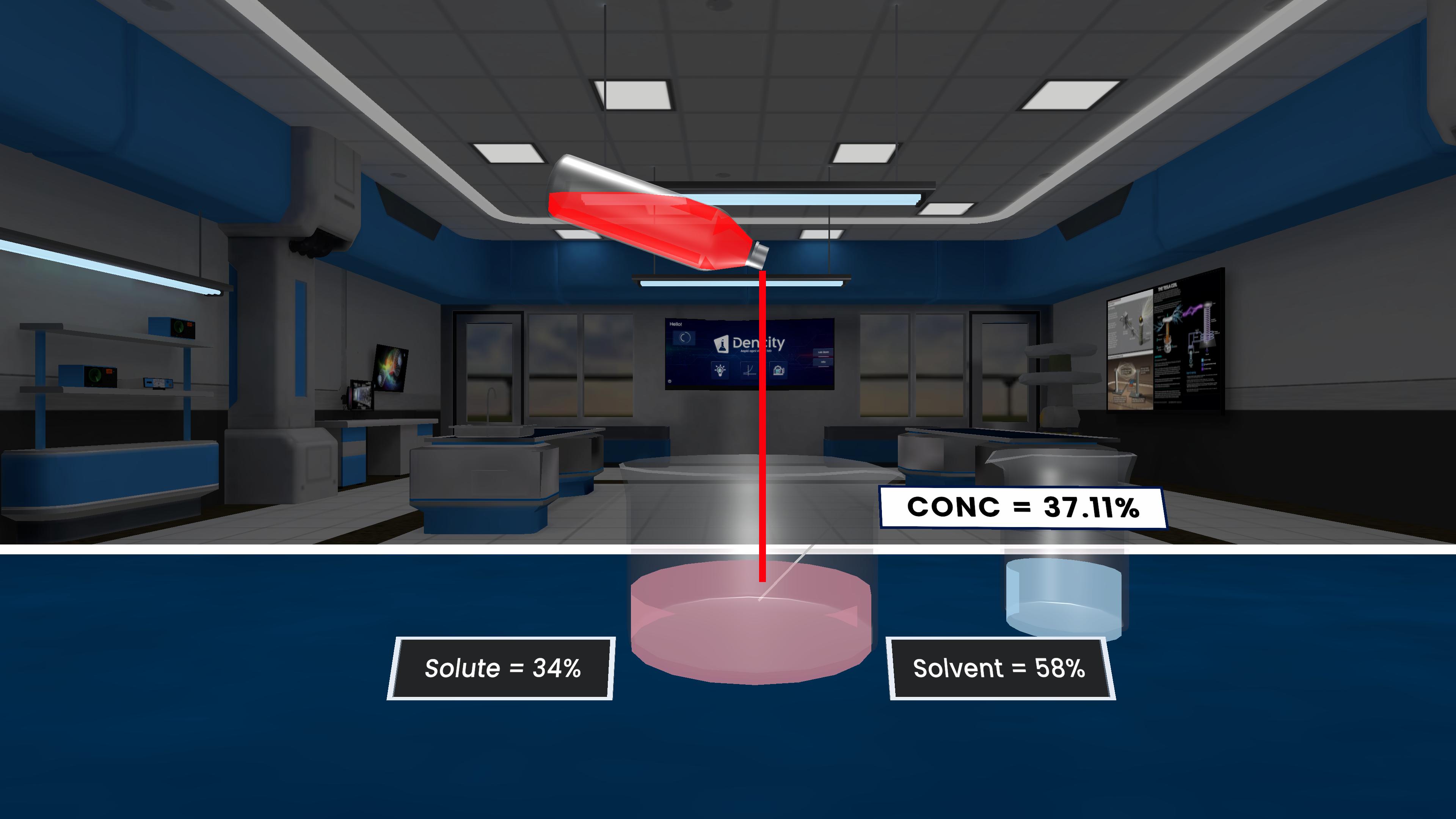
This experiment explains concentration in a solution. By adding different amounts of solute to a fixed amount of solvent, it shows that more solute makes the solution more concentrated, while less solute makes it dilute.
Experiment description
This experiment explains concentration in a solution. By adding different amounts of solute to a fixed amount of solvent, it shows that more solute makes the solution more concentrated, while less solute makes it dilute.
Keep browsing related experiments from the same category.

Chemistry
This experiment demonstrates how adding salt lowers the freezing point of water. Two beakers, one with pure water and the other with salt solution, ar...

Chemistry
Elevation of Boiling Point: This experiment demonstrates that adding salt increases the boiling point of water. Two beakers, one with pure water and o...

Chemistry
This experiment demonstrates how a saturated solution is formed. Salt is added to water and stirred until it dissolves. More salt is added gradually, ...