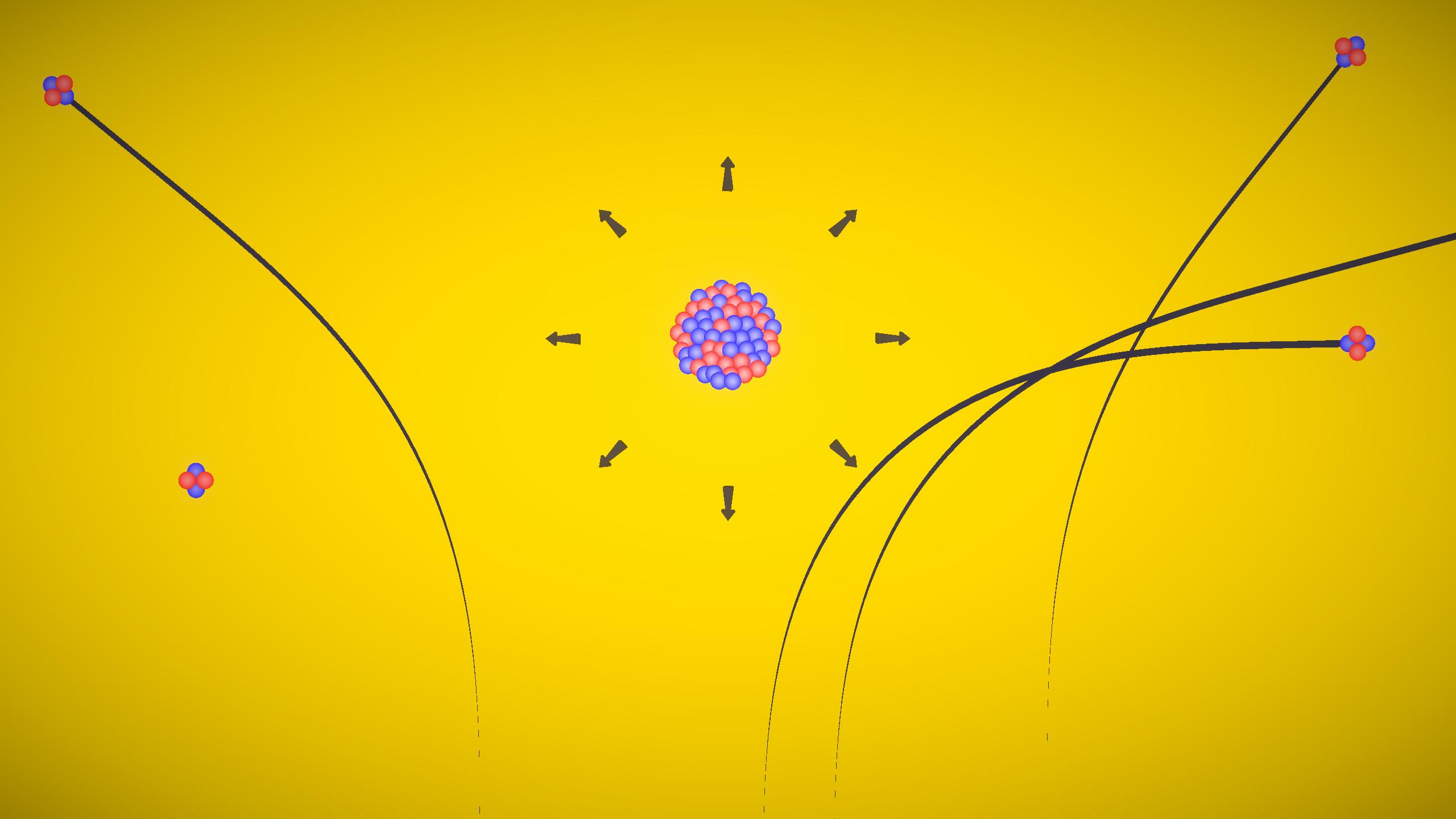
Experiment description
This experiment demonstrates Rutherford's alpha particle scattering. Alpha particles are directed toward a thin gold foil, and their scattering is observed due to electrostatic repulsion from the positively charged nucleus. The paths of the particles help explain the structure of the atom and the presence of a small, dense nucleus.