Experiment description
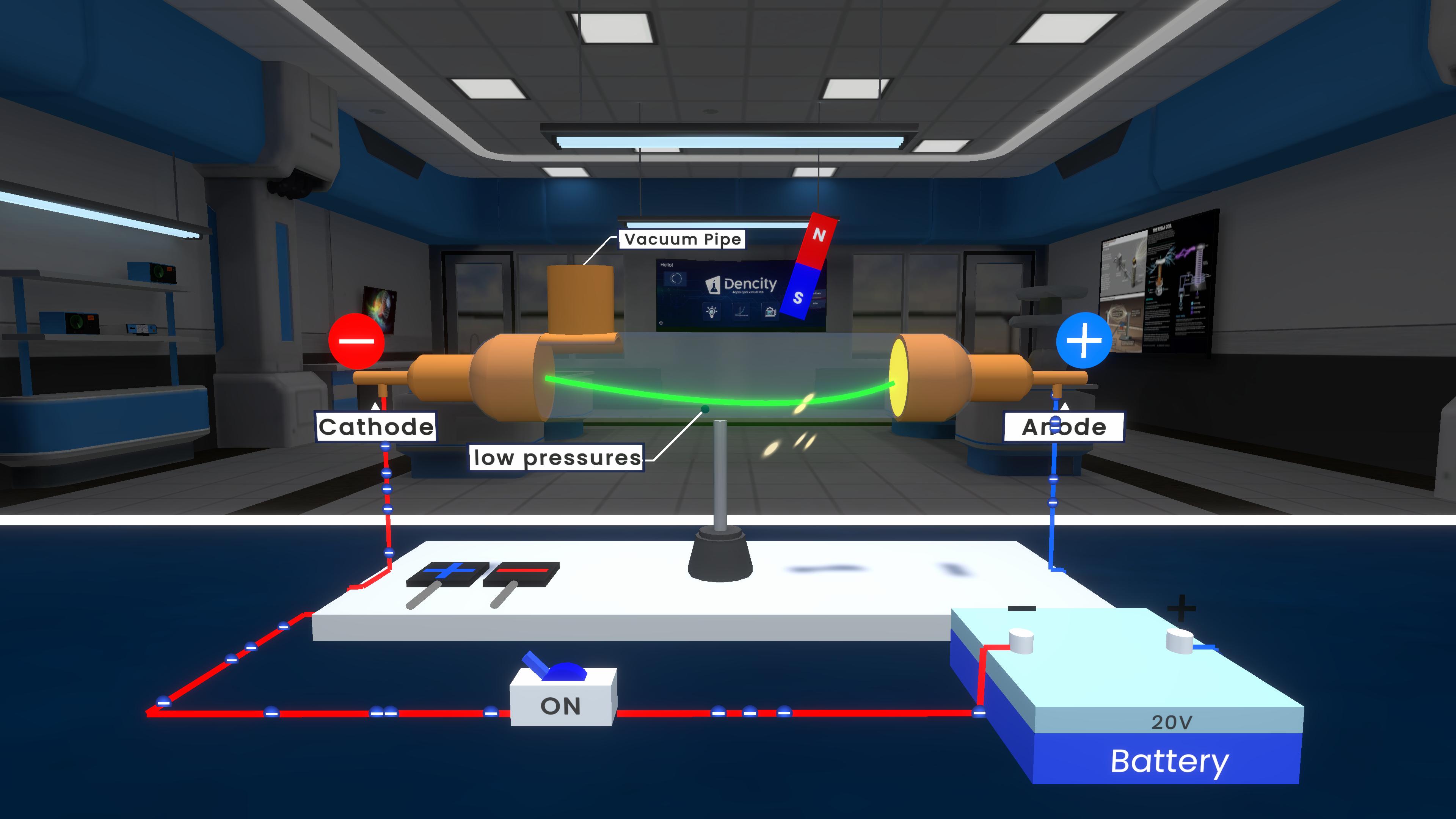
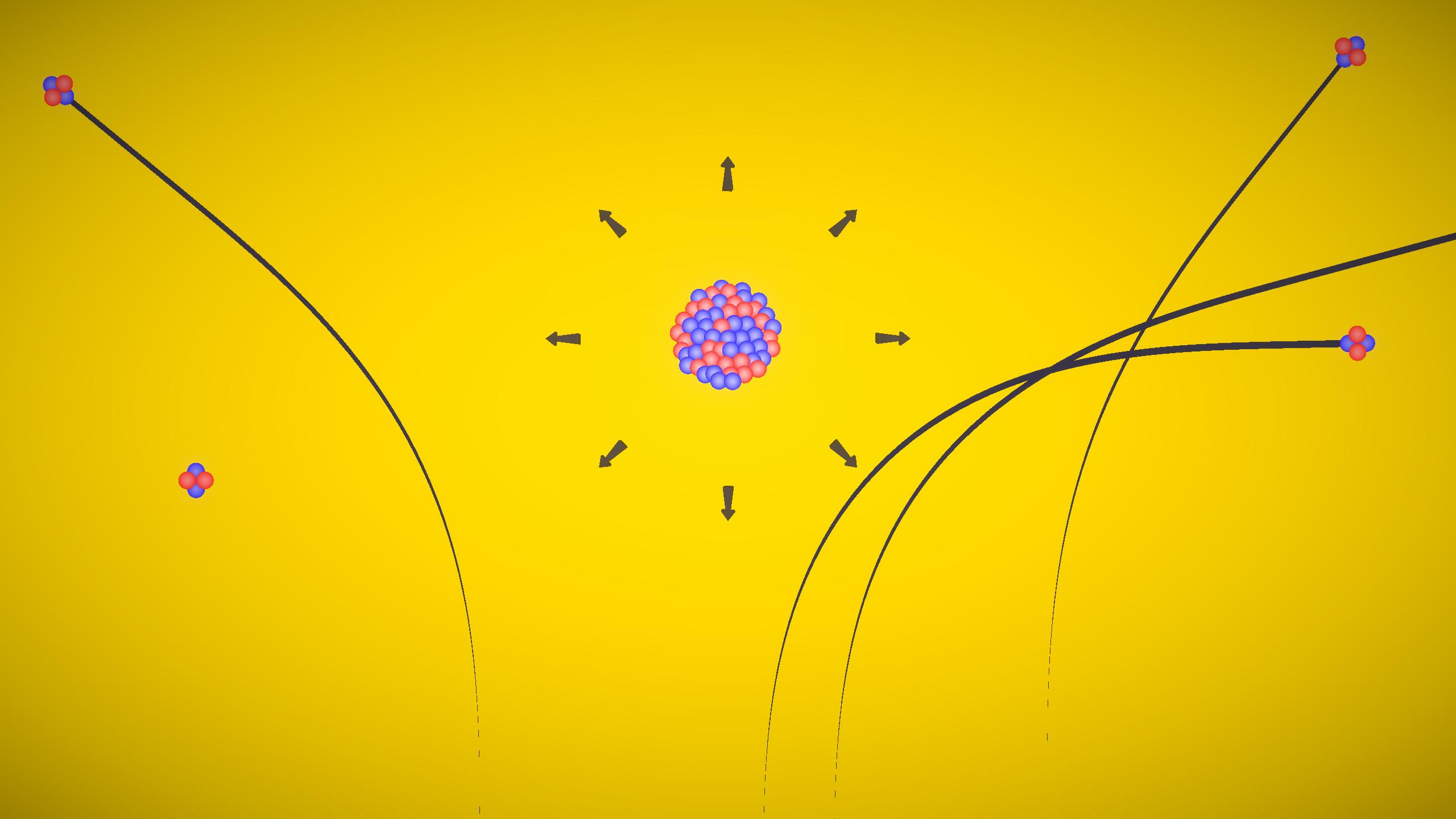
This experiment demonstrates cathode rays using a discharge tube. When high voltage is applied in a low-pressure gas tube, rays travel from the cathode to the anode in straight lines. These rays produce fluorescence and get deflected by electric and magnetic fields, proving they are made of negatively charged particles called electrons.