Experiment description
This experiment demonstrates Lewis structures by showing how atoms share or hold valence electrons to form molecules, using dots for lone pairs and lines for bonds, and explaining stability through the octet and duet rules.
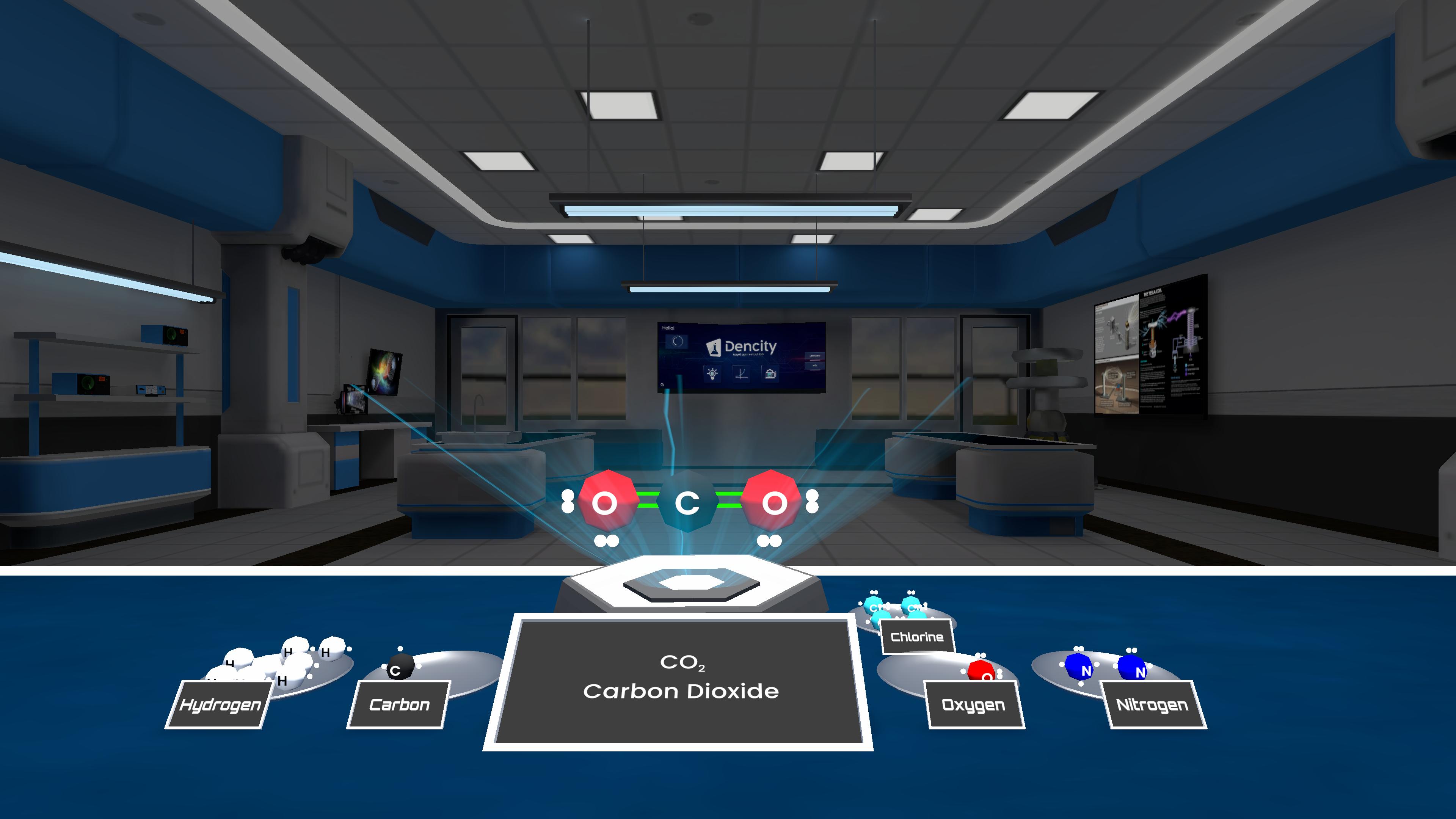
Experiment description
This experiment demonstrates Lewis structures by showing how atoms share or hold valence electrons to form molecules, using dots for lone pairs and lines for bonds, and explaining stability through the octet and duet rules.
Keep browsing related experiments from the same category.
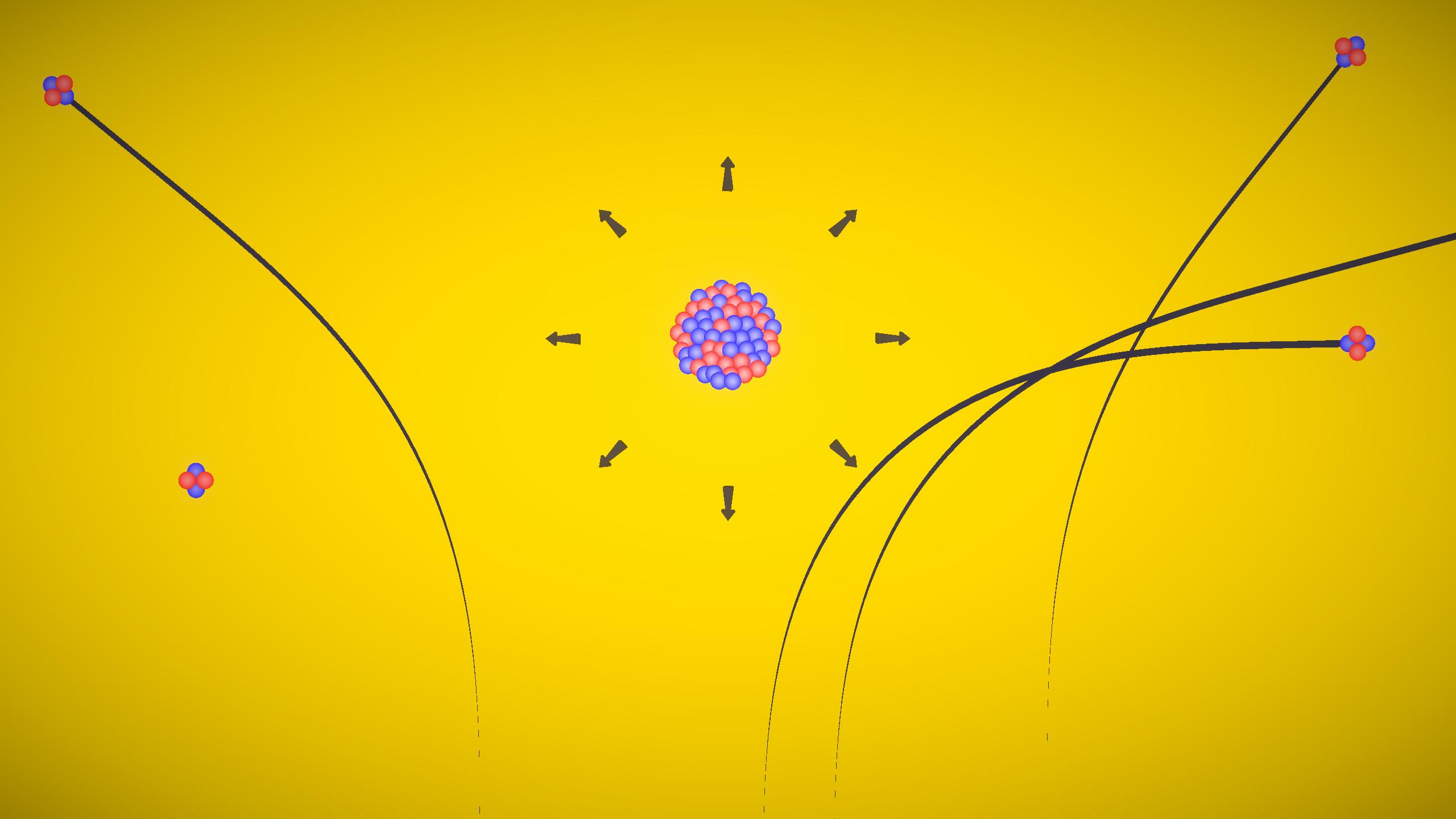
Physics
This experiment demonstrates Rutherford's alpha particle scattering. Alpha particles are directed toward a thin gold foil, and their scattering is obs...
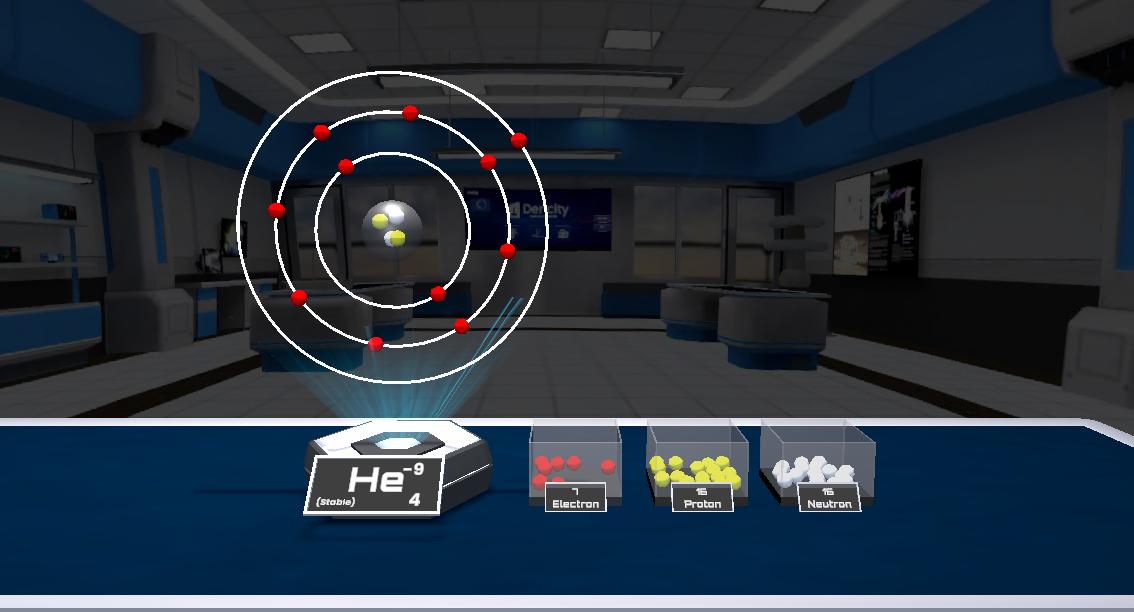
Physics
This experiment demonstrates how atoms are formed using protons, neutrons, and electrons. By adding or removing these particles, different elements an...

Physics
This experiment demonstrates Bohr's atomic model. Electrons can be given energy or made to release energy to move between the first, second, and third...