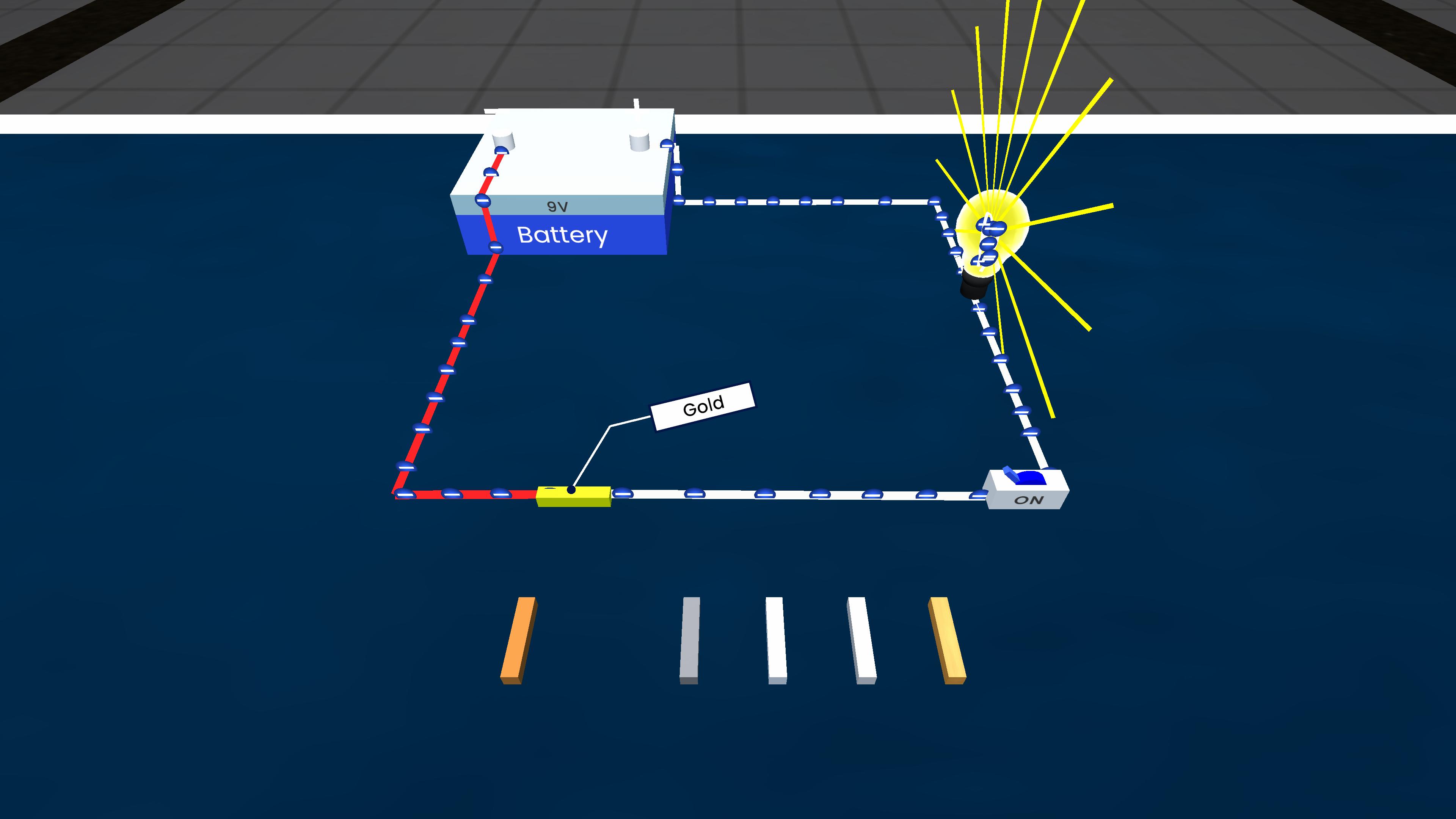
Experiment description
This experiment demonstrates displacement reactions between metals and solutions of other metal salts. When an iron nail is placed in copper sulphate solution, iron displaces copper, showing that iron is more reactive. When copper is placed in iron sulphate solution, no reaction occurs, indicating that copper is less reactive. This helps explain the reactivity series of metals.


![Effect of Steam on a Metal [Metals react with Water]](/Concept%20Panel%20Images/EffectOfSteamOnMetal_Image.jpg)