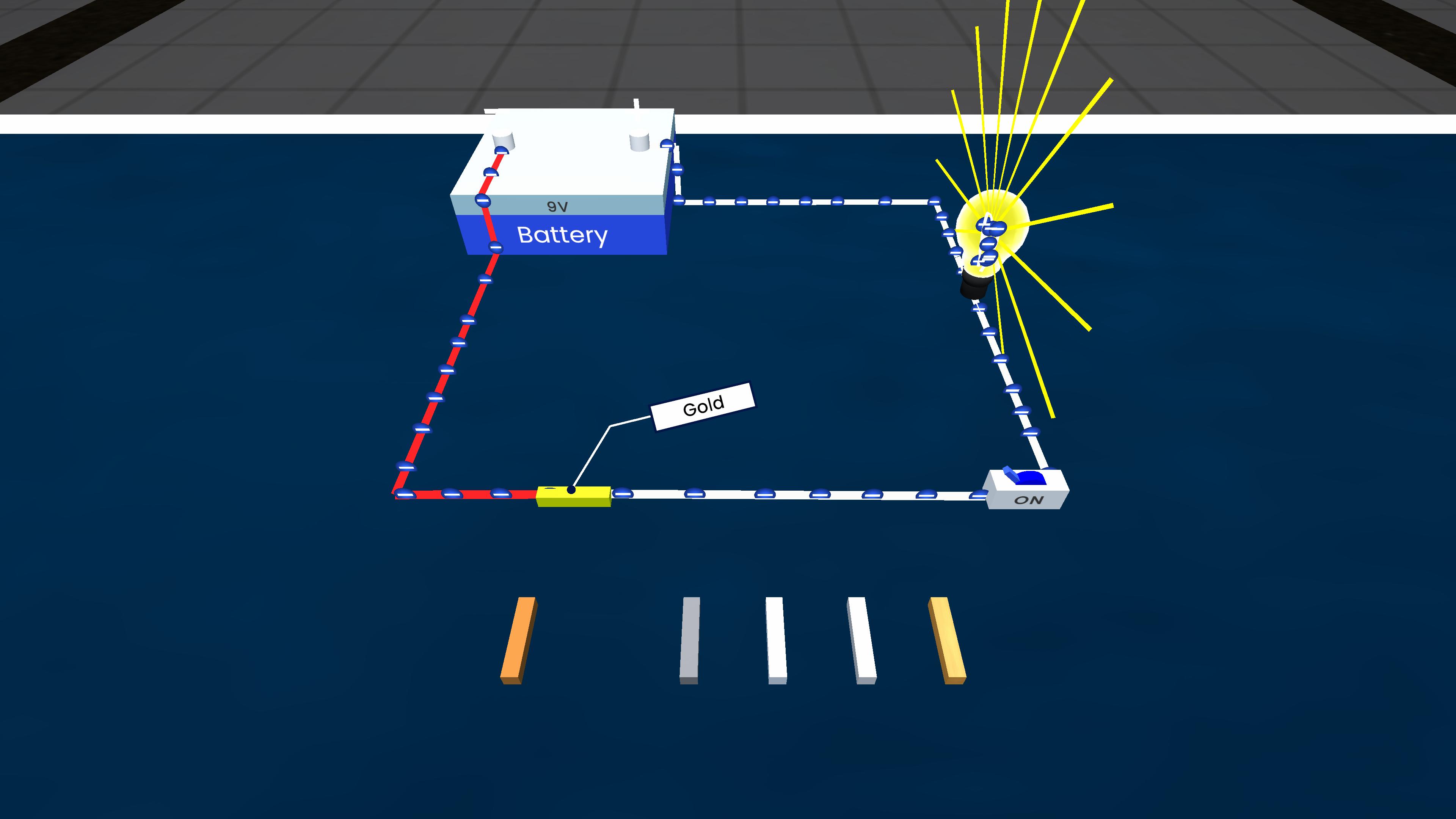
Experiment description
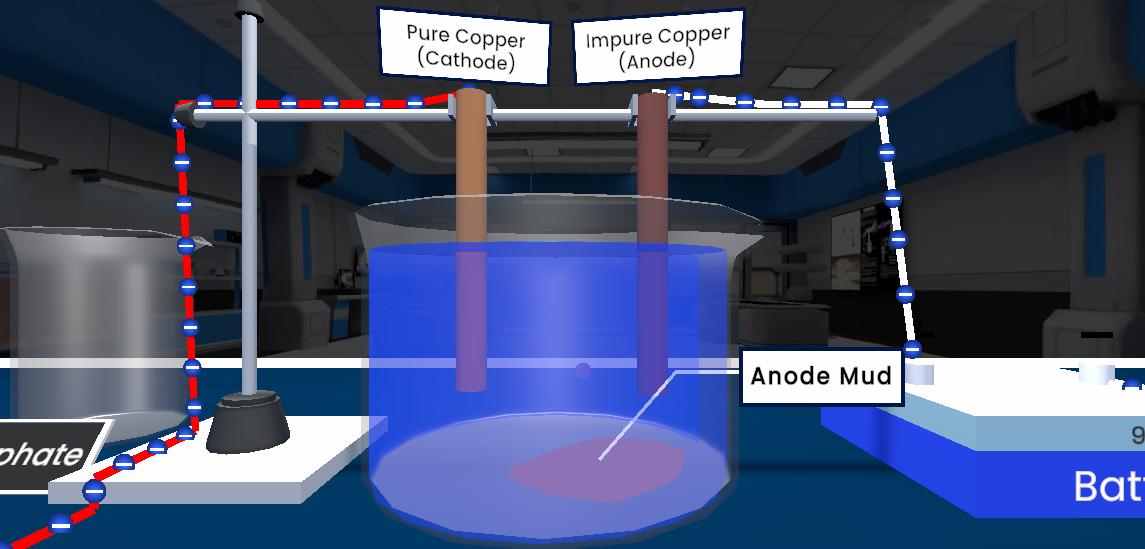
This experiment demonstrates the refining of metals using electrolytic refining. An impure metal is taken as the anode and a pure metal strip as the cathode, both placed in a solution of the metal's salt. When electric current is passed, pure metal from the anode dissolves and gets deposited on the cathode, while impurities either remain in the solution or settle at the bottom as anode mud.

![Effect of Steam on a Metal [Metals react with Water]](/Concept%20Panel%20Images/EffectOfSteamOnMetal_Image.jpg)